This is how the Clear & Simple Reference Tables Guides were born. I called it Bonding with the Reference Tables. I sold thousands of copies across New York State over a short period of time! The terrific feedback from teachers and their requests of "the same for Chemistry!" encouraged me to create the Chemistry reference tables guide. I named it Unearthing the Reference Tables. When I arrived back home, I finished the task I had begun. Three hours and 4 tables later, my laptop died. But I did not realize just how much there was to explain on each table until I got down to it. I had originally intended to make a brief, 4-5 page booklet. So, I took out my laptop and my Earth Science reference tables and began to work on a reference tables guide. Reference Tables for Physical Setting/Chemistry 2011 Edition : 9: 1: P eriod : 1.  
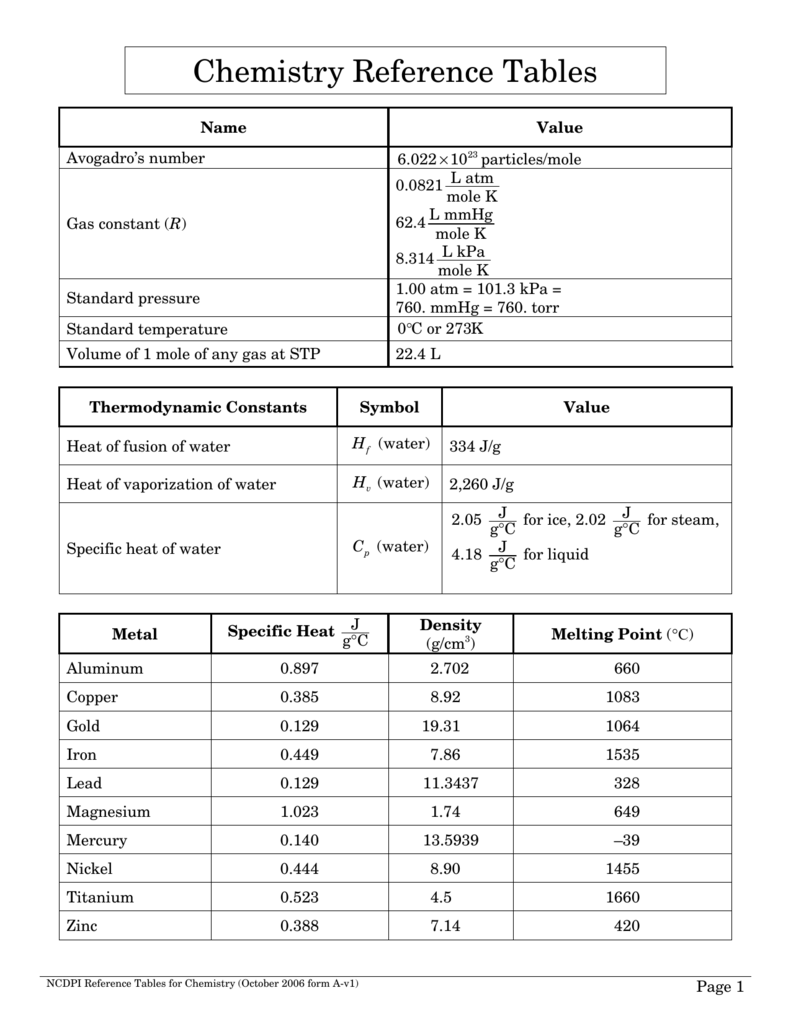
The Reference Tables are also used on the New York State Regents Exams. Reference Tables for Physical Setting/CHEMISTRY 2011 Edition Table A. The booklets are frequently used during classes, tests, and lab assignments. They contain important measurements, equations, maps, and identification tables. It seemed such a pity - the answers were right there in front of them during the test! The Reference Tables are an invaluable tool to the high school science student. I had noticed on tests that, for some reason, many of my students tended to get reference tables-based questions wrong. It was almost June, so I decided to start putting together a review booklet for my students. g H 2O, must be used as a conversion factor to calculate the maximum amount of solute, ammonium iodide, NH 4I, that can dissolve in the given amount of solvent, 75.0 grams of water, H 2O.Several years ago, a month before the regents, I was stuck in a car for the nine hour drive between New York and Toronto. In order to determine whether this solution is saturated or unsaturated, the solubility of ammonium iodide, NH 4I, which has a reported value of 172 g/100. Since the chemical formula for water, H 2O, is associated with the 100.-gram quantities in the denominators of the solubilities in Table 7.9.1, water, H 2O, is the solvent in this solution, and the remaining substance, ammonium iodide, NH 4I, is the solute, "by default." may require the use of the 2011 Edition Reference Tables for Physical Setting/Chemistry. Use the solubility information that is presented in Table 7.9.1 to determine whether the resultant solution is saturated or unsaturated, and calculate the amount of excess solute that remains undissolved in this solution.īefore a solubility limit can be applied as a conversion factor, each substance that is referenced in the given problem must first be classified as a solute or a solvent. A four-function or scienti c calculator and a copy of the 2011 Edition Reference Tables for Physical Setting/Chemistry must be available for you to use while taking this examination. NCDPI Reference Tables for Chemistry (2012) Chemistry Reference Tables Name Value Avogadro’s number 6.022 10u 23 particles/mole Gas constant (R) 0.0821 L atm mole K 62.4 L mmHg mole K 8.314 L kPa mole K Standard pressure 1.00 atm 101. You still need to know when and how to use each equations up the BRACKNELL Chemistry reference table, cannot to mention you wont having your calculator fork multiple-choice questions. 14155 Chemistry Reference Tables Name Value Avogadro’s number 6.022 10 23 particles/mole Gas constant (R) 0.0821 L atm mole K 62.4 L mmHg mole K 8.314 L kPa mole K Standard pressure 1.00 atm 101.3 kPa 760. However, dont think this means you get out of understanding the actual formulas themselves. \)Ī solution is prepared by mixing 129 grams of ammonium iodide and 75.0 grams of water at 20 degrees Celsius. NCDPI Reference Tables for Chemistry (2012) Stock No.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
